Wastewater treatment using chitosan and its derivatives: A mini review on latest developments
DOI:
https://doi.org/10.55779/nsb14411369Keywords:
chitosan, hydrogels, nanocomposites, water treatmentAbstract
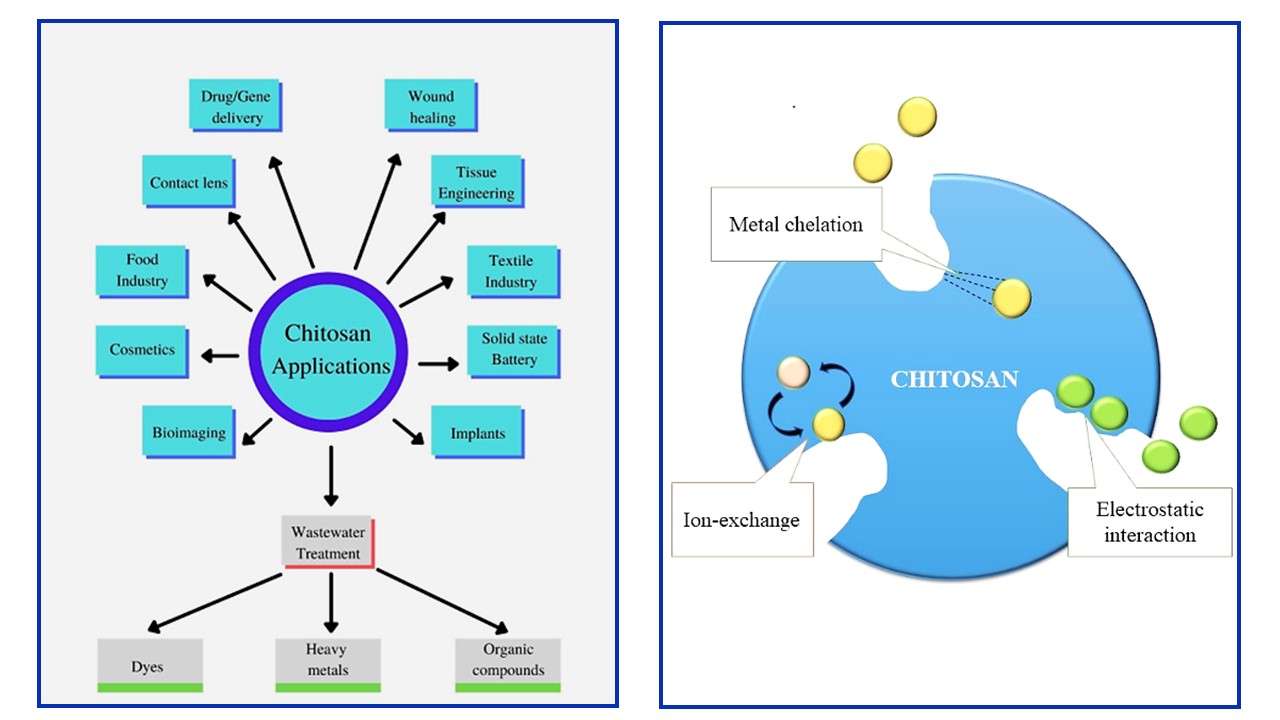
Effluents and contaminants released from the industries are needed to be treated before releasing them to water bodies. Most common effluents from these industrial wastes are organic compounds, dyes and heavy metals. Heavy metals and their associated anions, as well as organic material, have been separated from wastewaters in industries using a variety of methods. Adsorption is an effective method for water treatment as they are less energy consuming and cost effective. Biopolymers such as chitosan, cellulose, keratin are used for the process of adsorption as they are present abundantly and recyclable. Chitosan is a deacetylated product of chitin. Chitosan and its derivatives are extremely essential due to their abundant availability, low cost, environmental friendliness, and biodegradability and can be widely applied in wastewater treatment. -NH2 and -OH groups are present in chitosan and provide chitosan an opportunity to make physical and chemical modifications. Modifications of chitosan into hydrogels and nanocomposites provide wider applications in wastewater treatment.
Metrics
References
Abdelmouleh M, Boufi S, Belgacem MN, Duarte AP, Ben Salah A, Gandini A (2004). Modification of cellulosic fibres with functionalised silanes: development of surface properties. International Journal of Adhesion and Adhesives 24:43-54. https://doi.org/10.1016/S0143-7496(03)00099-X
Ahmad M, Yousaf M, Nasir A, Bhatti IA, Mahmood A, Fang X, Jian X, Kalantar-Zadeh K, Mahmood N (2019). Porous Eleocharis@MnPE layered hybrid for synergistic adsorption and catalytic biodegradation of toxic Azo dyes from industrial wastewater. Environmental Science & Technology 53:2161-2170. https://doi.org/10.1021/acs.est.8b05866
Aliabadi M, Irani M, Ismaeili J, Najafzadeh S (2014). Design and evaluation of chitosan/hydroxyapatite composite nanofiber membrane for the removal of heavy metal ions from aqueous solution. Journal of the Taiwan Institute of Chemical Engineers 45:518-526. https://doi.org/10.1016/j.jtice.2013.04.016
Al-Sherbini A-SA, Ghannam HEA, El-Ghanam GMA, El-Ella AA, Youssef AM (2019). Utilization of chitosan/Ag bionanocomposites as eco-friendly photocatalytic reactor for Bactericidal effect and heavy metals removal. Heliyon 5:e01980. https://doi.org/10.1016/j.heliyon.2019.e01980
Bai B, Xu X, Li C, Xing J, Wang H, Suo Y (2018). Magnetic Fe3O4@chitosan carbon microbeads: removal of doxycycline from aqueous solutions through a fixed bed via sequential adsorption and heterogeneous fenton-like regeneration. Journal of Nanomaterials 2018:1-14. https://doi.org/10.1155/2018/5296410.
Barakat MA (2011). New trends in removing heavy metals from industrial wastewater. Arabian Journal of Chemistry 4:361-377. https://doi.org/10.1016/j.arabjc.2010.07.019
Beheshti H, Irani M, Hosseini L, Rahimi A, Aliabadi M (2016). Removal of Cr (VI) from aqueous solutions using chitosan/MWCNT/Fe 3 O 4 composite nanofibers-batch and column studies. Chemical Engineering Journal 284:557-564. https://doi.org/10.1016/j.cej.2015.08.158.
Bhatnagar A, Sillanpää M (2009). Applications of chitin- and chitosan-derivatives for the detoxification of water and wastewater - A short review. Advances in Colloid and Interface Science 152:26-38. https://doi.org/10.1016/j.cis.2009.09.003.
Chang M-Y, Juang R-S (2004). Adsorption of tannic acid, humic acid, and dyes from water using the composite of chitosan and activated clay. Journal of Colloid and Interface Science 278:18-25. https://doi.org/10.1016/j.jcis.2004.05.029
Chauhan A, Sillu D, Agnihotri S (2019). Removal of pharmaceutical contaminants in wastewater using nanomaterials: a comprehensive review. Current Drug Metabolism 20:483-505. https://doi.org/10.2174/1389200220666181127104812
Cheng SY, Show P-L, Juan JC, Chang J-S, Lau BF, Lai SH, Ng EP, Yian HC, Ling TC (2021). Landfill leachate wastewater treatment to facilitate resource recovery by a coagulation-flocculation process via hydrogen bond. Chemosphere 262:127829. https://doi.org/10.1016/j.chemosphere.2020.127829
Cheung WH, Szeto YS, McKay G (2009). Enhancing the adsorption capacities of acid dyes by chitosan nano particles. Bioresource Technology 100:1143-1148. https://doi.org/10.1016/j.biortech.2008.07.071
Couto CF, Lange LC, Amaral MCS (2018). A critical review on membrane separation processes applied to remove pharmaceutically active compounds from water and wastewater. Journal of Water Process Engineering 26:156-175. https://doi.org/10.1016/j.jwpe.2018.10.010.
Crini G, Gimbert F, Robert C, Martel B, Adam O, Morin-Crini N, De Giorgi F, Badot P-M (2008). The removal of Basic Blue 3 from aqueous solutions by chitosan-based adsorbent: Batch studies. Journal of Hazardous Materials 153:96-106. https://doi.org/10.1016/j.jhazmat.2007.08.025
Crini G, Lichtfouse E, Wilson LD, Morin-Crini N (2018). Adsorption-oriented processes using conventional and non-conventional adsorbents for wastewater treatment. Environmental Chemistry for a Sustainable World 23-71. https://doi.org/10.1007/978-3-319-92111-2_2
Darvishi Cheshmeh Soltani R, Khataee AR, Safari M, Joo SW (2013). Preparation of bio-silica/chitosan nanocomposite for adsorption of a textile dye in aqueous solutions. International Biodeterioration & Biodegradation 85:383–391 https://doi.org/10.1016/j.ibiod.2013.09.004.
Dergunov SA, Mun GA (2009). γ-irradiated chitosan-polyvinyl pyrrolidone hydrogels as pH-sensitive protein delivery system. Radiation Physics and Chemistry 78:65-68. https://doi.org/10.1016/j.radphyschem.2008.07.003.
Desbrières J, Guibal E (2018). Chitosan for wastewater treatment. Polymer International 67:7-14. https://doi.org/10.1002/pi.5464.
Doshi B, Repo E, Heiskanen JP, Sirviö JA, Sillanpää M (2017). Effectiveness of N,O-carboxymethyl chitosan on destabilization of Marine Diesel, Diesel and Marine-2T oil for oil spill treatment. Carbohydrate Polymers 167:326-336. https://doi.org/10.1016/j.carbpol.2017.03.064.
Fu F, Wang Q (2011). Removal of heavy metal ions from wastewaters: A review. Journal of Environmental Management 92:407-418. https://doi.org/10.1016/j.jenvman.2010.11.011.
Fu Y, Xiao C (2017). A facile physical approach to make chitosan soluble in acid-free water. International Journal of Biological Macromolecules 103:575-580. https://doi.org/10.1016/j.ijbiomac.2017.05.066
Haider S, Park S-Y (2009). Preparation of the electrospun chitosan nanofibers and their applications to the adsorption of Cu(II) and Pb(II) ions from an aqueous solution. Journal of Membrane Science 328:90-96. https://doi.org/10.1016/j.memsci.2008.11.046.
Hameed BH (2007). Equilibrium and kinetics studies of 2,4,6-trichlorophenol adsorption onto activated clay. Colloids and Surfaces A: Physicochemical and Engineering Aspects 307:45-52. https://doi.org/10.1016/j.colsurfa.2007.05.002.
Hoseini MHM, Sadeghi S, Azizi M, Pouriran R (2020). Immunomodulatory activities of chitin and chitosan microparticles. Handbook of Chitin and Chitosan 609-639. https://doi.org/10.1016/b978-0-12-817966-6.00019-4.
Hu D, Lian Z, Xian H, Jiang R, Wang N, Weng Y, Peng X, Wang S, Ouyang X-K (2020). Adsorption of Pb(II) from aqueous solution by polyacrylic acid grafted magnetic chitosan nanocomposite. International Journal of Biological Macromolecules 154:1537-1547. https://doi.org/10.1016/j.ijbiomac.2019.11.038
Hu ZG, Zhang J, Chan WL, Szeto YS (2006). The sorption of acid dye onto chitosan nanoparticles. Polymer 47:5838-5842. https://doi.org/10.1016/j.polymer.2006.05.071.
Igberase E, Osifo P, Ofomaja A (2014). The adsorption of copper (II) ions by polyaniline graft chitosan beads from aqueous solution: Equilibrium, kinetic and desorption studies. Journal of Environmental Chemical Engineering 2:362-369. https://doi.org/10.1016/j.jece.2014.01.008.
Islam MM, Shahruzzaman M, Biswas S, Nurus Sakib M, Rashid TU (2020). Chitosan based bioactive materials in tissue engineering applications-A review. Bioactive Materials 5:164-183. https://doi.org/10.1016/j.bioactmat.2020.01.012
Jiang R, Fu Y-Q, Zhu H-Y, Yao J, Xiao L (2012). Removal of methyl orange from aqueous solutions by magnetic maghemite/chitosan nanocomposite films: Adsorption kinetics and equilibrium. Journal of Applied Polymer Science 125:E540-E549. https://doi.org/10.1002/app.37003.
Kalia S, Avérous L (2011). Biopolymers: Biomedical and Environmental Applications. John Wiley & Sons.
Kalyani P, Hemalatha KPJ (2016). Biosorption of heavy metals in the environment-a review paper. International Journal of Current Research and Academic Review 4:66-74. https://doi.org/10.20546/ijcrar.2016.411.011.
Karimi-Maleh H, Ayati A, Davoodi R, Tanhaei B, Karimi F, Malekmohammadi S, Orooji Y, Fu L, Sillanpää M (2021). Recent advances in using of chitosan-based adsorbents for removal of pharmaceutical contaminants: A review. Journal of Cleaner Production 291:125880. https://doi.org/10.1016/j.jclepro.2021.125880.
Kaur S, Dhillon GS (2014). The versatile biopolymer chitosan: potential sources, evaluation of extraction methods and applications. Critical Reviews in Microbiology 40:155-175. https://doi.org/10.3109/1040841x.2013.770385.
Kayalvizhi K, Alhaji NMI, Saravanakkumar D, Beer Mohamed S, Kaviyarasu K, Ayeshamariam A, … Elshikh MS (2022). Adsorption of copper and nickel by using sawdust chitosan nanocomposite beads – A kinetic and thermodynamic study. Environmental Research 203:111814. https://doi.org/10.1016/j.envres.2021.111814
Kekes T, Tzia C (2020). Adsorption of indigo carmine on functional chitosan and β-cyclodextrin/chitosan beads: Equilibrium, kinetics and mechanism studies. Journal of Environmental Management 262:110372. https://doi.org/10.1016/j.jenvman.2020.110372
Keshvardoostchokami M, Babaei S, Piri F, Zamani A (2017). Nitrate removal from aqueous solutions by ZnO nanoparticles and chitosan-polystyrene–Zn nanocomposite: Kinetic, isotherm, batch and fixed-bed studies. International Journal of Biological Macromolecules 101:922-930. https://doi.org/10.1016/j.ijbiomac.2017.03.162.
Kluczka J, Gnus M, Kazek-Kęsik A, Dudek G (2018). Zirconium-chitosan hydrogel beads for removal of boron from aqueous solutions. Polymer 150:109-118. https://doi.org/10.1016/j.polymer.2018.07.010.
Li P, Wang Y, Peng Z, She MF, Kong L (2010). Physichemical property and morphology of 5-fluorouracil loaded chitosan nanoparticles. 2010 International Conference on Nanoscience and Nanotechnology https://doi.org/10.1109/iconn.2010.6045203.
Linghu WS, Wang C (2014). Adsorption of heavy metal ions from aqueous solution by chitosan. Advanced Materials Research 881:570-573. https://doi.org/10.4028/www.scientific.net/AMR.881-883.570
Liu J, Pu H, Zhang X, Xiao L, Kan J, Jin C (2018). Effects of ascorbate and hydroxyl radical degradations on the structural, physicochemical, antioxidant and film forming properties of chitosan. International Journal of Biological Macromolecules 114:1086-1093. https://doi.org/10.1016/j.ijbiomac.2018.04.021
Liu X, Hu Q, Fang Z, Zhang X, Zhang B (2009). Magnetic chitosan nanocomposites: a useful recyclable tool for heavy metal ion removal. Langmuir: The ACS Journal of Surfaces and Colloids 25:3-8. https://doi.org/10.1021/la802754t
Mazhar SN, Ahmad S (2020). Assessment of water quality pollution indices and distribution of heavy metals in drinking water in Ramganga aquifer, Bareilly District Uttar Pradesh, India. Groundwater for Sustainable Development 10:100304. https://doi.org/10.1016/j.gsd.2019.100304
Menon AK, Haechler I, Kaur S, Lubner S, Prasher RS (2020). Enhanced solar evaporation using a photo-thermal umbrella for wastewater management. Nature Sustainability 3:144-151. https://doi.org/10.1038/s41893-019-0445-5
Miklos DB, Remy C, Jekel M, Linden KG, Drewes JE, Hübner U (2018). Evaluation of advanced oxidation processes for water and wastewater treatment - A critical review. Water Research 139:118-131. https://doi.org/10.1016/j.watres.2018.03.042
Mohammadzadeh Pakdel P, Peighambardoust SJ (2018). Review on recent progress in chitosan-based hydrogels for wastewater treatment application. Carbohydrate Polymers 201:264-279. https://doi.org/10.1016/j.carbpol.2018.08.070
Muzzarelli RAA, Lough C, Emanuelli M (1987). The molecular weight of chitosans studied by laser light-scattering. Carbohydrate Research 164:433-442. https://doi.org/10.1016/0008-6215(87)80146-5.
Nagarpita MV, Roy P, Shruthi SB, Sailaja RRN (2017). Synthesis and swelling characteristics of chitosan and CMC grafted sodium acrylate-co-acrylamide using modified nanoclay and examining its efficacy for removal of dyes. International Journal of Biological Macromolecules 102:1226-1240. https://doi.org/10.1016/j.ijbiomac.2017.04.099
Ngah WSW, Wan Ngah WS, Fatinathan S (2008). Adsorption of Cu(II) ions in aqueous solution using chitosan beads, chitosan–GLA beads and chitosan–alginate beads. Chemical Engineering Journal 143:62-72. https://doi.org/10.1016/j.cej.2007.12.006
Ngah WSW, Wan Ngah WS, Teong LC, Hanafiah MAK (2011). Adsorption of dyes and heavy metal ions by chitosan composites: A review. Carbohydrate Polymers 83:1446-1456. https://doi.org/10.1016/j.carbpol.2010.11.004
Nguyen LM, Nguyen TTH (2019). Enhanced heavy metals biosorption using chemically modified chitosan coated microwave activated sugarcane baggage ash composite biosorbents. SN Applied Sciences 1. https://doi.org/10.1007/s42452-019-1607-9.
Nippatla N, Philip L (2019). Electrocoagulation-floatation assisted pulsed power plasma technology for the complete mineralization of potentially toxic dyes and real textile wastewater. Process Safety and Environmental Protection 125:143-156. https://doi.org/10.1016/j.psep.2019.03.012
Olivera S, Muralidhara HB, Venkatesh K, Guna VK, Gopalakrishna K, Kumar KY (2016). Potential applications of cellulose and chitosan nanoparticles/composites in wastewater treatment: A review. Carbohydrate Polymers 153:600-618. https://doi.org/10.1016/j.carbpol.2016.08.017
Qi C, Zhao L, Lin Y, Wu D (2018a). Graphene oxide/chitosan sponge as a novel filtering material for the removal of dye from water. Journal of Colloid and Interface Science 517:18-27. https://doi.org/10.1016/j.jcis.2018.01.089
Qi L, Xu Z (2004). Lead sorption from aqueous solutions on chitosan nanoparticles. Colloids and Surfaces A: Physicochemical and Engineering Aspects 251:183-190. https://doi.org/10.1016/j.colsurfa.2004.10.010
Qi X, Wu L, Su T, Zhang J, Dong W (2018b). Polysaccharide-based cationic hydrogels for dye adsorption. Colloids Surf B Biointerfaces 170:364-372. https://doi.org/10.1016/j.colsurfb.2018.06.036
Rabea EI (2014). In vitro assessment of antimicrobial property of O-(phenoxyacetic) chitosan compounds on plant pathogens. Journal of Chitin and Chitosan Science 2:293-298. https://doi.org/10.1166/jcc.2014.1081
Ramírez-Estrada A, Mena-Cervantes VY, Fuentes-García J, Vazquez-Arenas J, Palma-Goyes R, Flores-Vela AI, … Hernández Altamirano R (2018). Cr(III) removal from synthetic and real tanning effluents using an electro-precipitation method. Journal of Environmental Chemical Engineering 6:1219-1225. https://doi.org/10.1016/j.jece.2018.01.038
Rao TP, Prasada Rao T, Kala R, Daniel S (2006). Metal ion-imprinted polymers—Novel materials for selective recognition of inorganics. Analytica Chimica Acta 578:105-116. https://doi.org/10.1016/j.aca.2006.06.065.
Rinaudo M, Milas M, Desbrières J (2020). Characterization and solution properties of chitosan and chitosan derivatives. Applications of Chitin and Chitosan 89-102. https://doi.org/10.1201/9781003072812
Saha S, Zubair M, Khosa MA, Song S, Ullah A (2019). Keratin and chitosan biosorbents for wastewater treatment: a review. Journal of Polymers and the Environment 27:1389-1403. https://doi.org/10.1007/s10924-019-01439-6.
Salehi E, Daraei P, Shamsabadi AA (2016). A review on chitosan-based adsorptive membranes. Carbohydrate Polymers 152:419-432. https://doi.org/10.1016/j.carbpol.2016.07.033
Salehi R, Arami M, Mahmoodi NM, Bahrami H, Khorramfar S (2010). Novel biocompatible composite (Chitosan–zinc oxide nanoparticle): Preparation, characterization and dye adsorption properties. Colloids and Surfaces B: Biointerfaces 80:86-93. https://doi.org/10.1016/j.colsurfb.2010.05.039
Sarode S, Upadhyay P, Khosa MA, Mak T, Shakir A, Song S, Ullah A (2019). Overview of wastewater treatment methods with special focus on biopolymer chitin-chitosan. International Journal of Biological Macromolecules 121:1086-1100. https://doi.org/10.1016/j.ijbiomac.2018.10.089
Shariatinia Z (2019). Pharmaceutical applications of chitosan. Advances in Colloid and Interface Science 263:131-194. https://doi.org/10.1016/j.cis.2018.11.008
Sheth Y, Dharaskar S, Khalid M, Sonawane S (2021). An environment friendly approach for heavy metal removal from industrial wastewater using chitosan based biosorbent: A review. Sustainable Energy Technologies and Assessments 43:100951. https://doi.org/10.1016/j.seta.2020.100951
Shukla SK, Mishra AK, Arotiba OA, Mamba BB (2013). Chitosan-based nanomaterials: a state-of-the-art review. Int J Biol Macromol 59:46-58.
Sun K, Gao B, Zhang Z, Zhang G, Liu X, Zhao Y, Xing B (2010). Sorption of endocrine disrupting chemicals by condensed organic matter in soils and sediments. Chemosphere 80:70-715. https://doi.org/10.1016/j.chemosphere.2010.05.028
Thirugnanasambandham K, Sivakumar V, Prakash M (2014). Treatment of egg processing industry effluent using chitosan as an adsorbent. Journal of the Serbian Chemical Society 79:743-757. https://doi.org/10.2298/JSC130201053T
Tsai W-T, Hsu H-C, Su T-Y, Lin K-Y, Lin C-M (2006). Adsorption characteristics of bisphenol-A in aqueous solutions onto hydrophobic zeolite. Journal of Colloid and Interface Science 299:513-519. https://doi.org/10.1016/j.jcis.2006.02.034
Vakili M, Rafatullah M, Salamatinia B, Abdullah AZ, Ibrahim MH, Tan KB, Gholami Z, Amouzgar P (2014). Application of chitosan and its derivatives as adsorbents for dye removal from water and wastewater: a review. Carbohydrate Polymers 113:115-130. https://doi.org/10.1016/j.carbpol.2014.07.007
Vidal RRL, Moraes JS (2019). Removal of organic pollutants from wastewater using chitosan: a literature review. International Journal of Environmental Science and Technology 16:1741-1754. https://doi.org/10.1007/s13762-018-2061-8
Volpin F, Fons E, Chekli L, Kim JE, Jang A, Shon HK (2018). Hybrid forward osmosis-reverse osmosis for wastewater reuse and seawater desalination: Understanding the optimal feed solution to minimise fouling. Process Safety and Environmental Protection 117:523-532. https://doi.org/10.1016/j.psep.2018.05.006
Wong YC, Szeto YS, Cheung WH, McKay G (2003). Equilibrium studies for acid dye adsorption onto chitosan. Langmuir 19:7888-7894. https://doi.org/10.1021/la030064y
Wu ACM (1988). Determination of molecular-weight distribution of chitosan by high-performance liquid chromatography. Methods in Enzymology 447-452. https://doi.org/10.1016/0076-6879(88)61055-X
Yuwei C, Jianlong W (2011). Preparation and characterization of magnetic chitosan nanoparticles and its application for Cu(II) removal. Chemical Engineering Journal 168:286-292. https://doi.org/10.1016/j.cej.2011.01.006
ZabihiSahebi A, Koushkbaghi S, Pishnamazi M, Askari A, Khosravi R, Irani M (2019). Synthesis of cellulose acetate/chitosan/SWCNT/Fe3O4/TiO2 composite nanofibers for the removal of Cr(VI), As(V), Methylene blue and Congo red from aqueous solutions. International Journal of Biological Macromolecules 140:1296-1304. https://doi.org/10.1016/j.ijbiomac.2019.08.214
Zahedifar M, Seyedi N, Shafiei S, Basij M (2021). Surface-modified magnetic biochar: Highly efficient adsorbents for removal of Pb(ΙΙ) and Cd(ΙΙ). Materials Chemistry and Physics 271:124860. https://doi.org/10.1016/j.matchemphys.2021.124860
Zhang X, Ye C, Pi K, Huang J, Xia M, Gerson AR (2019). Sustainable treatment of desulfurization wastewater by ion exchange and bipolar membrane electrodialysis hybrid technology. Separation and Purification Technology 211:330-339. https://doi.org/10.1016/j.seppur.2018.10.003
Zubair M, Arshad M, Ullah A (2020). Chitosan-based materials for water and wastewater treatment. Handbook of Chitin and Chitosan 773-809. https://doi.org/10.1016/B978-0-12-817966-6.00025-X
Downloads
Published
How to Cite
Issue
Section
License
Papers published in Notulae Scientia Biologicae are Open-Access, distributed under the terms and conditions of the Creative Commons Attribution License.
© Articles by the authors; licensee SMTCT, Cluj-Napoca, Romania. The journal allows the author(s) to hold the copyright/to retain publishing rights without restriction.
License:
Open Access Journal - the journal offers free, immediate, and unrestricted access to peer-reviewed research and scholarly work, due SMTCT supports to increase the visibility, accessibility and reputation of the researchers, regardless of geography and their budgets. Users are allowed to read, download, copy, distribute, print, search, or link to the full texts of the articles, or use them for any other lawful purpose, without asking prior permission from the publisher or the author.













.png)















