Possible ameliorative effects of hydromethanol extract of Thymus vulgaris on cadmium induced hepatorenal toxicity in rats
DOI:
https://doi.org/10.15835/nsb12310765Keywords:
antioxidant; cadmium; kidney; liver; Thymus vulgarisAbstract
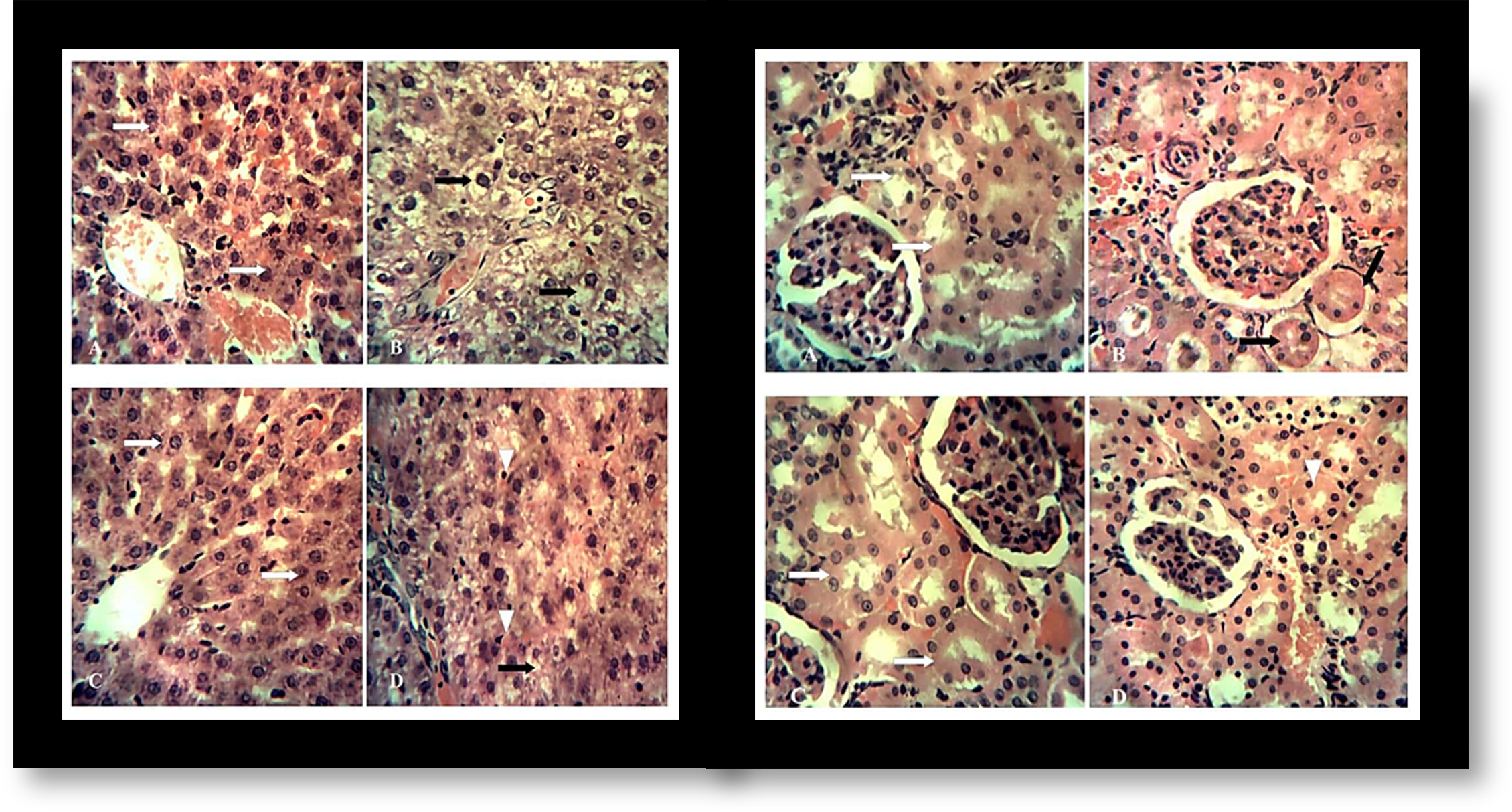
This study evaluated the possible ameliorative effect of hydromethanol extract of Thymus vulgaris on hepatorenal toxicity induced by cadmium in male rats. The experimental animals were divided into four groups and treated as follows: A (control - 0.5ml of 2% tween 80 in distilled water per os) for 3 weeks and a single subcutaneous dose of phosphate buffered saline, B (single subcutaneous dose of cadmium in phosphate buffered saline at 3 mg/kg); C (500 mg/kg extract per os daily for 3 weeks) and D (single subcutaneous dose of cadmium in phosphate buffered saline at 3 mg/kg + 500 mg/kg extract per os daily for 3 weeks). Cadmium administration resulted in suppression of erythrocyte count, hemoglobin concentration, packed cell volume, an elevated total leucocyte count with associated neutrophilia which improved with extract administration. Levels of serum alanine aminotransferase, aspartate aminotransferase and alkaline phosphatase, creatinine and total bilirubin concentration increased with decrease in total serum protein and albumin in cadmium treated group B compared to control group A, group C (extract only) and cadmium plus extract treated group D. Cadmium led to a reduction in catalase and superoxide dismutase activities with increase in the level of malondialdehyde. However, co-administration of extract with cadmium in group D reduced lipid peroxidation and oxidative stress induced by cadmium. Histopathological examination of cadmium treated groups showed moderate vacuolar degeneration in the liver and degeneration of the kidney tubules which were ameliorated following co-administration with extract. This study shows that Thymus vulgaris extract has a potential protective effect against cadmium induced hepato-renal injury through the suppression of oxidative stress.
Metrics
References
Hadwan MH (2018). Simple spectrophotometric assay for measuring catalase activity in biological tissues. BMC Biochemistry 19(7):1-8. https://doi.org/10.1186/s12858-018-0097-5
Bancroft JD, Gamble M (2002). Theory and practice of histological techniques. Edinburgh: Churchill Livingstone.
Blass KG, Thiebert RJ, Lam LK (1974). A study of the mechanism of the Jaffe reaction. Journal of Clinical Chemistry and Clinical Biochemistry 12(7):336-343. https://doi.org/10.1515/cclm.1974.12.7.336
Rahimzadeh RM, Rahimzadeh RM, Kazemi S, Moghadamnia AA (2017). Cadmium toxicity and treatment: An update. Caspian Journal of Internal Medicine 8(3):135-145. https://doi.org/10.22088/cjim.8.3.135
Thrall MA, Weiser MG (2002). Hematology. In: Hendrix CM (Ed). Laboratory Procedures for Veterinary Technicians, 4th edn. Mosby, Missouri pp 29-74.
Dan G, Lall SB, Rao DN (2000). Humoral and cell mediated immune response to cadmium in mice. Drug and Chemical Toxicology 23(2):349-360. https://doi.org/10.1081/DCT-100100120
Daud D, Fekery NFM, Hashim N (2017). Reproductive health of rats with benign prostatic hyperplasia following Cosmos caudatus ethanolic extracts consumption. Journal of Applied Pharmaceutical Science 7(6):202-205. https://doi.org/10.7324/JAPS.2017.70630
Descotes J (1992). Immunotoxicology of cadmium. IARC Scientific Publications 118:385-390.
Donmez HH, Donmez N, Kısadere I, Undag I (2019). Protective effect of quercetin on some hematological parameters in rats exposed to cadmium. Biotechnic and Histochemistry 94(5):381-386. https://doi.org/10.1080/10520295.2019.1574027
Doumas BT, Watson WA, Biggs HG (1997). Albumin standards and the measurement of serum albumin with bromcresol green. 1971. Clinica Chimica Acta 258(1):21-30. https://doi.org/10.1016/s0009-8981(96)06447-9
Dzobo K, Naik YS (2013). Effect of selenium on cadmium-induced oxidative stress and esterase activity in rat organs. South African Journal of Science 109(5-6):1-8. https://doi.org/10.1590/sajs.2013/965
El-Refaiy AI, Eissa FI (2013). Histopathology and cytotoxicity as biomarkers in treated rats with cadmium and some therapeutic agents. Saudi Journal of Biological Sciences 20(3):265-280. https://doi.org/10.1016/j.sjbs.2013.02.004
El-Habit OH, Abdel Moneim AE (2014). Testing the genotoxicity, cytotoxicity and oxidative stress of cadmium and nickel and their additive effect in male mice. Biological Trace Element Research 159(1-3):364-372.
https://doi.org/10.1007/s12011-014-0016-6
El-Nekeety AA, Mohamed SR, Hathout AS, Hassan NS, Aly SE, Abdel-Wahhab MA (2011). Antioxidant properties of Thymus vulgaris oil against aflatoxin-induce oxidative stress in male rats. Toxicon 57(7-8):984-991. https://doi.org/10.1016/j.toxicon.2011.03.021
Gulcin I, Beydemir S (2013). Phenolic compounds as antioxidants: carbonic anhydrase isoenzymes inhibitors. Mini-Reviews in Medicinal Chemistry 13(3):408-430. https://doi.org/10.2174/1389557511313030009
Gülçin I (2012). Antioxidant activity of food constituents: an overview. Archives of Toxicology 86(3):345-391. https://doi.org/10.1007/s00204-011-0774-2
Haidry MT, Malik A (2016). Therapeutic effects of Cassia angustifolia in cadmium induced hepatotoxicity assay conducted in male albino rats. Bulletin of Environmental Studies 1(2):39-42.
Horiguchi H, Oguma E (2016). Acute exposure to cadmium induces prolonged neutrophilia along with delayed induction of granulocyte colony-stimulating factor in the livers of mice. Archives of Toxicology 90(12):3005-3015. https://doi.org/10.1007/s00204-016-1661-7
Huff J, Lunn RM,Waalkes MP, Tomatis L, Infante PF (2007). Cadmium-induced cancers in animals and in humans. International Journal of Occupational and Environmental Health 13(2):202-212. https://doi.org/10.1179/oeh.2007.13.2.202
IARC (2012). Evaluation of carcinogenic risks to humans: International Agency for Research on Cancer. Lyon, France. Retrieved 2020 January 1 from https://monographs.iarc.fr/wp content/uploads/2018/06/mono100E.pdf
Higgins T, Beutler E, Doumas BT (2008). Measurement of haemoglobin in blood. In: Burtis CA, Ashwood ER, Bruns DE (eds), Tietz Fundamentals of Clinical Chemistry, 6th edn. Sanders Elsevier, Missouri, pp 524-525.
Kumar A, Pandey R, Siddiqi NJ, Sharma B (2019). Oxidative stress biomarkers of cadmium toxicity in mammalian systems and their distinct ameliorative strategy. Journal of Applied Biotechnology and Bioengineering 6(3):126-135.
Johnson AM (2008). Aminoacids and proteins. In: Burtis CA, Ashwood ER, Bruns DE (Eds). Tietz Fundamentals of Clinical Chemistry, Saunders Elsevier, Missouri pp 206-316.
Matović V, Buha A, Ðukić-Ćosić D, Bulat Z (2015). Insight into the oxidative stress induced by lead and/or cadmium in blood, liver and kidneys. Food and Chemical Toxicology 78:130-140. https://doi.org/10.1016/S0278-6915 (15)00074-5
Milica GP, Milos MM, Branka IO, Zorica SS (2017). Antioxidative and haematoprotective activity of coenzyme Q10 and vitamin E against cadmium-induced oxidative stress in Wistar rats. Toxicology and Industrial Health 33(10):746-756. https://doi.org/10.1177/0748233717725480
Mladenovic J, Ognjanovic B, Ðordevic N, Matić M, Knežević V, Štajn A, Saičić Z (2014). Protective effects of estradiol against cadmium induced changes in blood parameters and oxidative damage in rats. Archives of Industrial Hygiene and Toxicology 65(1):37-46. https://doi.org/10.2478/10004-1254-65-2014-2405
Mouihs JM, Thevenod F (2010). New perspectives in cadmium toxicity: an introduction. Biometals 23(5):763-768. https://doi.org/10.1007/s10534-010-9365-6
NRC (2011). National Research Council Committee for the Update of the Guide for the Care and Use of Laboratory Animals. Guide for the Care and Use of Laboratory Animals. 8th edition. Washington (DC): National Academies Press (US).
Nishikimi M, Appaji N, Yagi K (1972). The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. Biochemical Biophysical Research Communication 46(2):849-854. https://doi.org/10.1016/S0006-291X(72)80218-3
Nordberg GF, Fowler B, Nordberg M (2014). Handbook on the toxicology of metals. 4th Ed. Elsevier: Academic Press, New York.
Ocaña A, Reglero G (2012). Effects of thyme extract oils (from Thymus vulgaris, Thymus zygis, and Thymus hyemalis) on cytokine production and gene expression of oxLDL-Stimulated THP-1-Macrophages. Journal of Obesity 2012:104706. https://doi.org/10.1155/2012/104706
OECD (2002). Test No. 423: Acute Oral toxicity - Acute Toxic Class Method, Organization for Economic Cooperation and Development Guidelines for the Testing of Chemicals, Section 4, OECD Publishing (in Paris). Retrieved 2019 April 12 from https://doi.org/10.1787/9789264071001-en
Ohkawa H, Ohishi N, Yagi K (1979). Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Analytical Biochemistry 95(2):351-358. https://doi.org/10.1016/0003-2697(79)90738-3
Ognjanovic BI, Pavlovic SZ, Maletic SD, Zikić RV, Stajn AS, Radojicić RM, … Petrović VM (2003). Protective influence of vitamin E on antioxidant defense system in the blood of rats treated with cadmium. Physiological Research 52(5):563-570.
Oyinloye BE, Ajiboye BO, Ojo OA, Nwozo SO, Kappo AP (2016). Cardio-protective and antioxidant influence of aqueous extracts from Sesamum indicum seeds on oxidative stress induced by cadmium in wistar rats. Pharmacognosy Magazine 12(46):170-174.
Ponnusamy M, Pari L (2011). Protective role of diallyl tetrasulfide on cadmium-induced testicular damage in adult rats: A biochemical and histological study. Toxicology and Industrial Health 27(5):407-416. https://doi.org/10.1177/0748233710387633
Pruett SB, Hebert P, Lapointe J, Reagan ML, Kawabata TT (2007). Characterization of the action of drug-induced stress responses on the immune system: Evaluations of biomarkers for drug-induced stress in rats. Journal of Immunotoxicology 4(1):25-38. https://doi.org/10.1080/15476910601115150
Pushpavalli G, Veeramani C, Pugalendi KV (2010). Influence of chrysin on hepatic marker enzymes and lipid profile against D-galactosamine-induced hepatotoxicity rats. Food and Chemical Toxicology 48(6):1654-1659. https://doi.org/10.1016/j.fct.2010.03.040
Colville J (2002). Blood chemistry. In: Hendrix CM (Ed). Laboratory procedures for Veterinary Technicians, 4th edn. Mosby, St Louis pp 75-103.
Sathiavelu J, Senapathy GJ, Devaraj R, Namasivayam N (2009). Hepatoprotective effect of chrysin on prooxidant‐antioxidant status during ethanol‐induced toxicity in female albino rats. The Journal of Pharmacy and Pharmacology 61(6):809-817. https://doi.org/10.1211/jpp/61.06.0015
Schulte S, Mengel K, Gatke U, Friedberg KD (1994). No influence of cadmium on the production of specific antibodies in mice. Toxicology 93(2-3):263-268. https://doi.org/10.1016/0300-483x(94)90083-3
Satyal P, Murray BL, McFeeters RL, Setzer WN (2016). Essential oil characterization of Thymus vulgaris from various geographical locations. Foods 5(4):70. https://doi.org/10.3390/foods5040070
Suh YJ, Lee JE, Lee DH, Yi HG, Lee MH, Kim CS, … Kim SK (2016). Prevalence and relationships of iron deficiency anemia with blood cadmium and vitamin levels in Korean women. Journal of Korean Medical Science 31(1):25-32. https://doi.org/10.3346/jkms.2016.31.1.25
Vigo E, Cepeda A, Gualillo O, Perez-Fernandez R (2004). In vitro anti-inflammatory effect of Eucalyptus globulus and Thymus vulgaris: nitric oxide inhibition in J774A.1 murine macrophage. The Journal of Pharmacy and Pharmacology 56(2):257-263. https://doi.org/10.1211/0022357022665
Wojdylo A, Oszmianski J, Czemerys R (2007). Antioxidant activity and phenolic compounds in 32 selected herbs. Food Chemistry 105(3):940-949. https://doi.org/10.1016/j.foodchem.2007.04.038
Zarei L, Shahrooz R (2019). Protective effects of Cornus mas fruit extract on methotrexate-induced alterations in mice testicular tissue: Evidences for histochemical and histomorphometrical changes in an animal model study. Veterinary Research Forum 10 (4):307-313. https://doi.org/10.30466/vrf.2019.69516.1955
Downloads
Published
How to Cite
Issue
Section
License
Papers published in Notulae Scientia Biologicae are Open-Access, distributed under the terms and conditions of the Creative Commons Attribution License.
© Articles by the authors; licensee SMTCT, Cluj-Napoca, Romania. The journal allows the author(s) to hold the copyright/to retain publishing rights without restriction.
License:
Open Access Journal - the journal offers free, immediate, and unrestricted access to peer-reviewed research and scholarly work, due SMTCT supports to increase the visibility, accessibility and reputation of the researchers, regardless of geography and their budgets. Users are allowed to read, download, copy, distribute, print, search, or link to the full texts of the articles, or use them for any other lawful purpose, without asking prior permission from the publisher or the author.













.png)















