Preliminary screening of selected soils in Ilorin, North-central Nigeria for antibiotic-producing actinomycetes
DOI:
https://doi.org/10.15835/nsb12110544Keywords:
antibacterial; bioactive; inhibition; physicochemical; Streptomyces bottropensisAbstract
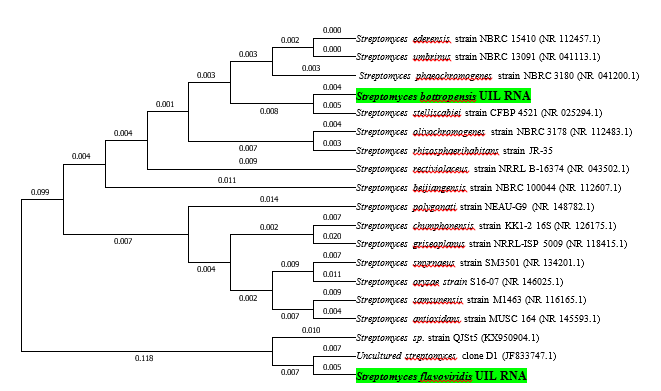
The present research aimed at screening various soils within Ilorin metropolis for antibiotic producing actinomycetes. The objectives of the study were to determine physiochemical parameters of soils, the occurrence of actinomycetes in soils, antibacterial potentials and identity of isolates. Soil parameters such as pH, temperature, moisture, organic matter and soil type were evaluated following standard procedures. Selective isolation to determine the occurrence of actinomycetes was performed by soil dilution using pour plate technique on starch casein agar. Preliminary antibacterial screening against 10 clinical test bacteria was performed using cross streak method. All isolates were initially identified based on morphological and biochemical characteristics, while the most bioactive isolates were further identified by molecular means. The soils were alkaline, with temperatures between 29 °C and 31 °C, moisture was in range of 0.72 ± 0.07c and 6.62 ± 0.42b. Highest organic matter content was 32.13 ± 0.20a with soil types mostly loamy and sandy loam. Ten actinomycetes (SM1 - SM10) were isolated, with the most frequently occurring isolate being SM3 and SM5 (16.7%). SM5 was the most active, inhibiting 9 out of 10 tests, with the highest inhibition against Staphylococcus aureus 25923 (24 mm ± 0.15a). All isolates were identified as Streptomyces by morphology and biochemical tests. Based on nucleotide similarity searches and phylogeny, two bioactive Streptomycetes were suggested as novel strains and thus named as Streptomyces bottropensis UIL RNA (SM5) and S. flavoviridis UIL RNA(SM7), which may serve as promising sources of antibiotics. Actinomycetes from Ilorin metropolis demonstrated broad spectrum of antibacterial activity against clinical test bacteria.
Metrics
References
Afifi MM, Atta HM, Bayoumi RA (2014). Microbiological studies on the production of antimicrobial agent by actinomycete isolated from Saudi Arabia. Research Journal of Microbiology 9:1-15.
Edwards SE, Morel CL, Busse R, Harbarth S (2018). Combatting antibiotic resistance together: how can we enlist the help of industry? Antibiotics 7(4):111.
Fawole MO, Oso BA (2007). Laboratory manual of microbiology (5th ed), Ibadan, Nigeria. Spectrum book limited.
George M, Anjumol A, George, G, Hatha AM (2012). Distribution and bioactive potential of soil actinomycetes from different ecological habitats. African Journal of Microbiology Research 6(10):2265-2271.
Ghorbani-Nasrabadi R, Greiner R, Alikhani HA, Hamedi J, Yakhchali B (2013). Distribution of actinomycetes in different soil ecosystems and effect of media composition on extracellular phosphatase activity. Journal of Soil Science and Plant Nutrition 13(1):223-236.
Gulve RM, Deshmukh AM (2012). Antimicrobial activity of the marine actinomycetes. International Multidisciplinary Research Journal 2(3):16-22.
Ismail S, Ban A, Racha A (2015). Testing of production of inhibitory bioactive compounds by soil streptomycetes as preliminary screening programs in UAE for anti-cancer and anti-bacterial drugs. International Journal of Current Microbiology and Applied Science 4:446-459.
Jacoby R, Peukert M, Succurro A, Koprivova A, Koprivova S (2017). The role of soil microorganisms in plant mineral nutrition-current knowledge and future directions. Frontier in Plant Science 8:1617.
Kong K, Schneper L, Mathee K (2010). Beta-lactam antibiotics: from antibiosis to resistance and bacteriology. Journal of Pathology, Microbology and Immunoogy 118:1-36.
Li B, Webster TJ (2018). Bacteria antibiotic resistance: new challenges and opportunities for implant-associated orthopedic infections. Journal of Orthopedic Research 36(1):22-32.
Makut DM, Owolewa AO (2011). Antibiotic-producing fungi present in the soil environment of Keffi Metroplolis, Nasarawa State, Nigeria. Trakia Journal of Science 9(2):33-39.
McCauley A, Jones C, Olson-Rutz K (2017). Soil pH and organic matter. Nutrient Management 8:4449-4448.
Muhammad AA, Mst NJ, Nigar S, Farhana R (2016). Effect of temperature, pH and NaCl on the isolates of actinomycetes from straw and compost samples from Savar, Dharka, Bangladesh. American Journal of Microbiology and Immunology 1(2):10-15.
Newman DJ (2016). Predominately uncultured microbes as sources of bioactive agents. Frontier in Microbiology 7:1832.
Sampò S, Bergero R, Buffa G, Luppi-Mosca AM (1997). Soil fungal communities in a young and an old Alnus viridis coenosis. Mycologia 89(6):837-845.
Sharma M, Dangi P, Choudhary M (2014). Actinomycetes: source, identification, and their applications. International Journal of Current Microbiology and Applied Science 3(2):801-832.
Valli S, Suvathi SS, Aysha OS, Nirmala P, Vinoth KP, Reena A (2012). Antimicrobial potential of Actinomycetes species isolated from marine environment. Asian Pacific Journal of Tropical Biomedicine 2(6):469-473.
Ventola CL (2015). The antibiotic resistance crisis. Pharmacy and Therapeutics 40(4):277.
Yan N, Marchsner P, Cao W, Zuo C, Qin W (2015). Influence of salinity and water content on oil microorganisms. International Soil and Water Conservation Research 3(4):316-323.
Downloads
Published
How to Cite
Issue
Section
License
Papers published in Notulae Scientia Biologicae are Open-Access, distributed under the terms and conditions of the Creative Commons Attribution License.
© Articles by the authors; licensee SMTCT, Cluj-Napoca, Romania. The journal allows the author(s) to hold the copyright/to retain publishing rights without restriction.
License:
Open Access Journal - the journal offers free, immediate, and unrestricted access to peer-reviewed research and scholarly work, due SMTCT supports to increase the visibility, accessibility and reputation of the researchers, regardless of geography and their budgets. Users are allowed to read, download, copy, distribute, print, search, or link to the full texts of the articles, or use them for any other lawful purpose, without asking prior permission from the publisher or the author.













.png)















